Topic: Electrolytic Cell
Electrolytic Cell
Which process requires energy to decompose a substance?
(1) electrolysis
(2) neutralization
(3) sublimation
(4) synthesis
Which device requires electrical energy to produce a chemical change?
(1) electrolytic cell
(2) salt bridge
(3) voltaic cell
(4) voltmeter
Which process requires energy for a nonspontaneous redox reaction to occur?
(1) deposition
(2) electrolysis
(3) alpha decay
(4) chromatography
Energy is required to produce a chemical change during
(1) chromatography
(2) electrolysis
(3) boiling
(4) melting
Given the diagram representing an incomplete electrochemical cell:
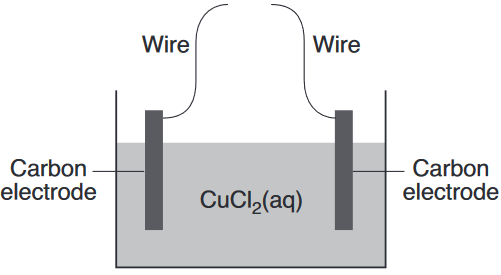
Solid copper will be deposited on one of the carbon electrodes when the wires are connected to
(1) each other
(2) a battery
(3) a switch
(4) a voltmeter
The electrolysis of brine, a concentrated aqueous sodium chloride solution, produces three important industrial chemicals: chlorine gas, hydrogen gas, and sodium hydroxide. The diagram and equation below represent a brine electrolysis cell. Before the battery is connected, the pH value of the brine solution is 7.0.
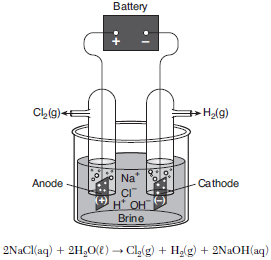
Explain, in terms of energy, why this cell is an electrolytic cell.
Allow 1 credit. Acceptable responses include, but are not limited to:
• Electrical energy is required to cause a non-spontaneous chemical change.
• The battery supplies the electricity needed for the reaction to occur.
A student develops the list shown below that includes laboratory equipment and materials for constructing a voltaic cell.

Identify one item of laboratory equipment required to build an electrolytic cell that is not included in the list.
Allow 1 credit. Acceptable responses include, but are not limited to:
• battery
• external power source
• source of electricity
Fossil fuels produce air pollution and may eventually be depleted. Scientists are researching ways to use hydrogen as an alternate fuel.
A device called an artificial leaf was invented to produce hydrogen and oxygen using sunlight and water. The artifical leaf is an electrochemical cell. Equations 1 and 2 below represent the reactions taking place in the leaf. Equation 3 represents a reaction of hydrogen when used as fuel.
Equation 1: 2H2O + energy from sunlight → O2 + 4H+ + 4e−
Equation 2: 4H+ + 4e− → 2H2
Equation 3: 2H2(g) + O2(g) → 2H2O(g) + energy
Explain, in terms of energy, why the artificial leaf is an electrolytic cell.
Allow 1 credit. Acceptable responses include, but are not limited to:
• Sunlight is used as an external power source for the cell.
• Sunlight is required to cause a nonspontaneous chemical change.
• Energy is required.
Metallic elements are obtained from their ores by reduction. Some metals, such as zinc, lead, iron, and copper, can be obtained by heating their oxides with carbon.
More active metals, such as aluminum, magnesium, and sodium, can not be reduced by carbon. These metals can be obtained by the electrolysis of their molten (melted) ores. The diagram below represents an incomplete cell for the electrolysis of molten NaCl. The equation below represents the reaction that occurs when the completed cell operates.

Identify one metal from the passage that is more active than carbon and one metal from the passage that is less active than carbon.
Allow 1 credit for identifying one metal from the passage that is more active than carbon and one metal from the passage that is less active than carbon.
• More active than carbon:
• ■ aluminum
• ■ Mg
• ■ Na
• Less active than carbon:
• ■ zinc
• ■ Pb
• ■ Fe
• ■ copper
The diagram below represents an operating electrolytic cell used to plate silver onto a nickel key. As the cell operates, oxidation occurs at the silver electrode and the mass of the silver electrode decreases.
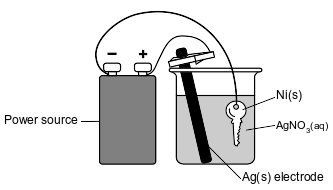
State the purpose of the power source in the cell.
Allow 1 credit. Acceptable responses include, but are not limited to:
• The cell requires electrical energy for the nonspontaneous reaction to occur.
• The power source causes some Ag(s) atoms to oxidize.
