Topic: Elements In The Periodic Table
Elements In The Periodic Table
The elements on the Periodic Table of the Elements are arranged in order of increasing
(1) atomic number
(2) mass number
(3) number of neutrons
(4) number of valence electrons
The elements on the Periodic Table of the Elements are arranged in order of increasing
(1) atomic mass
(2) formula mass
(3) atomic number
(4) oxidation number
On the Periodic Table, the number of protons in an atom of an element is indicated by its
(1) atomic mass
(2) atomic number
(3) selected oxidation states
(4) number of valence electrons
The arrangement of the elements from left to right in Period 4 on the Periodic Table is based on
(1) atomic mass
(2) atomic number
(3) the number of electron shells
(4) the number of oxidation states
The elements in Period 4 on the Periodic Table are arranged in order of increasing
(1) atomic radius
(2) atomic number
(3) number of valence electrons
(4) number of occupied shells of electrons
The elements on the Periodic Table are arranged in order of increasing
(1) mass number
(2) atomic number
(3) number of isotopes
(4) number of valence electrons
All elements on the modern Periodic Table are arranged in order of increasing
(1) atomic mass
(2) molar mass
(3) number of neutrons per atom
(4) number of protons per atom
The elements on the Periodic Table are arranged in order of increasing
(1) atomic mass
(2) atomic number
(3) molar mass
(4) oxidation number
The element in Group 14, Period 3, of the Periodic Table is classifi ed as a
(1) metal
(2) noble gas
(3) metalloid
(4) nonmetal
Which element is classifi ed as a metalloid?
(1) boron
(2) potassium
(3) sulfur
(4) xenon
Which element is malleable at STP?
(1) chlorine
(2) copper
(3) helium
(4) sulfur
Which element is classified as a metalloid?
(1) Te
(2) S
(3) Hg
(4) I
In the late 1800s, Dmitri Mendeleev developed a periodic table of the elements known at that time. Based on the pattern in his periodic table, he was able to predict properties of some elements that had not yet been discovered. Information about two of these elements is shown in the table below.
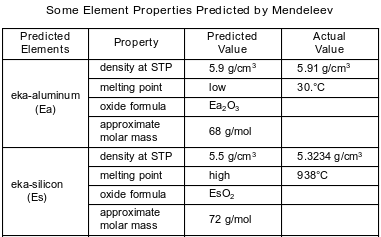
Identify the element that Mendeleev called eka-silicon, Es.
Allow 1 credit. Acceptable responses include, but are not limited to:
• Ge
• germanium
Many scientists made observations of the elements that led to the modern Periodic Table. In 1829, Dobereiner found groups of three elements that have similar properties and called each of these groups a triad. Dobereiner noticed a relationship between the atomic masses of the elements in each triad. Triad 1, shown in the table below, consists of sulfur, selenium, and tellurium. The middle element, selenium, has an atomic mass that is close to the sum of the atomic masses of sulfur and tellurium, divided by 2.
![]()
The other triads shown in the table below demonstrate the same mathematical relationship.
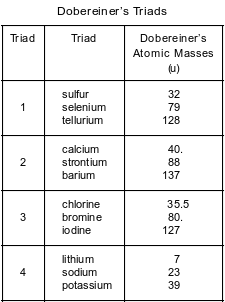
Show a numerical setup that demonstrates Dobereiner’s mathematical relationship for triad 2.
Allow 1 credit. Acceptable responses include, but are not limited to:
• 
Before atomic numbers were known, Mendeleev developed a classification system for the 63 elements known in 1872, using oxide formulas and atomic masses. He used an R in the oxide formulas to represent any element in each group. The atomic mass was listed in parentheses after the symbol of each element. A modified version of Mendeleev’s classification system is shown in the table below.
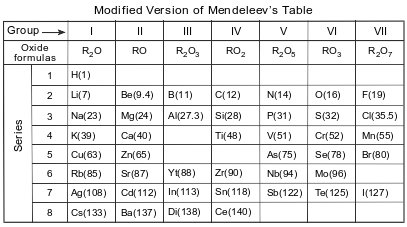
Identify one characteristic used by Mendeleev to develop his classification system of the elements.
Allow 1 credit. Acceptable responses include, but are not limited to:
• increasing atomic mass
• atomic mass
• oxide formulas
