Topic: Proton Neutron Electron
Proton Neutron Electron
In all atoms of bismuth, the number of electrons must equal the
(1) number of protons
(2) number of neutrons
(3) sum of the number of neutrons and protons
(4) difference between the number of neutrons and protons
The diagram below represents a particle traveling through an electric field.
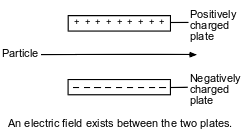
Which particle remains undeflected when passing through this electric field?
(1) proton
(2) electron
(3) neutron
(4) positron
Atoms are neutral because the number of
(1) protons equals the number of neutrons
(2) protons equals the number of electrons
(3) neutrons is greater than the number of protons
(4) neutrons is greater than the number of electrons
What is the net charge of an ion that has 11 protons, 10 electrons, and 12 neutrons?
(1) 1+
(2) 2+
(3) 1−
(4) 2−
The table below shows the number of protons, neutrons, and electrons in four ions.
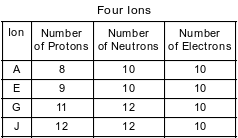
Which ion has a charge of 2−?
(1) A
(2) E
(3) G
(4) J
Which statement explains why a xenon atom is electrically neutral?
(1) The atom has fewer neutrons than electrons.
(2) The atom has more protons than electrons.
(3) The atom has the same number of neutrons and electrons.
(4) The atom has the same number of protons and electrons.
An ion that consists of 7 protons, 9 neutrons, and 10 electrons has a net charge of
(1) 2−
(2) 2+
(3) 3+
(4) 3−
What is the number of electrons in an atom that has 20 protons and 17 neutrons?
(1) 37
(2) 20
(3) 3
(4) 17
What is the number of electrons in an atom of scandium?
(1) 21
(2) 24
(3) 45
(4) 66
Which statement describes the charge of an electron and the charge of a proton?
(1) An electron and a proton both have a charge of +1.
(2) An electron and a proton both have a charge of −1.
(3) An electron has a charge of +1, and a proton has a charge of −1.
(4) An electron has a charge of −1, and a proton has a charge of +1.
What is the number of electrons in a potassium atom?
(1) 18
(2) 19
(3) 20
(4) 39
Which subatomic particles are paired with their charges?
(1) electron–positive, neutron–negative, proton–neutral
(2) electron–negative, neutron–neutral, proton–positive
(3) electron–negative, neutron–positive, proton–neutral
(4) electron–neutral, neutron–positive, proton–negative
Compared to an electron, which particle has a charge that is equal in magnitude but opposite in sign?
(1) an alpha particle
(2) a beta particle
(3) a neutron
(4) a proton
The atomic mass and natural abundance of the naturally occuring isotopes of hydrogen are shown in the table below.
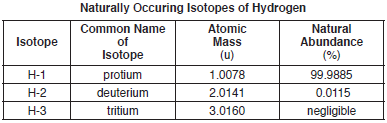
The isotope H-2, also called deuterium, is usually represented by the symbol “D.” Heavy water forms when deuterium reacts with oxygen, producing molecules of D2O.
Explain, in terms of subatomic particles, why atoms of H-1, H-2, and H-3 are each electrically neutral.
Allow 1 credit. Acceptable responses include, but are not limited to:
• The single proton in each nucleus has a charge of +1. The single electron in each atom has a charge of −1. The net charge is 0.
• Each atom has one proton and one electron.
• Each atom has an equal number of protons and electrons.
• The total charge of the subatomic particles is zero.
One fission reaction for U-235 is represented by the balanced nuclear equation below.
![]()
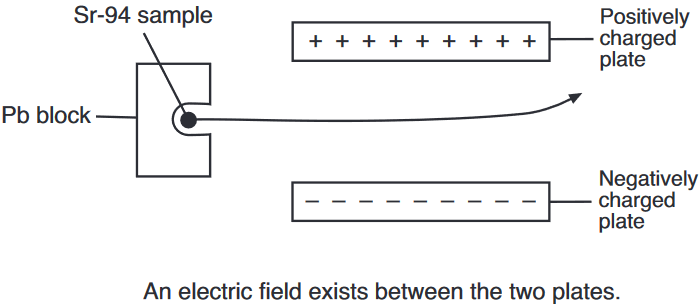
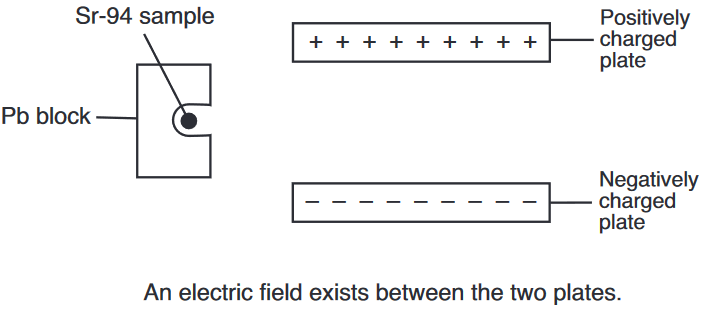
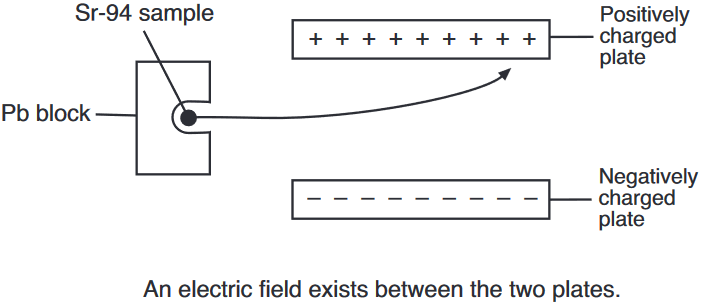
Both radioisotopes produced by this fission reaction undergo beta decay. The half-life of Xe-140 is 13.6 seconds and the half-life of Sr-94 is 1.25 minutes.
On the diagram below, draw an arrow to represent the path of an emitted beta particle in the electric field between two oppositely charged metal plates.
Allow 1 credit.
• Examples of 1-credit responses:
• 
•