Topic: Arrhenius Acids And Bases
Arrhenius Acids And Bases
Which compound is an Arrhenius base?
(1) HCl
(2) H3PO4
(3) Ca(OH)2
(4) CH3COOH
Which formula can represent hydrogen ions in an aqueous solution?
(1) OH−(aq)
(2) Hg22+(aq)
(3) H3O+(aq)
(4) NH4+(aq)
Which substance is an Arrhenius base?
(1) HNO3
(2) H2SO3
(3) Ca(OH)2
(4) CH3COOH
Which substance is an Arrhenius acid?
(1) H2
(2) HCl
(3) KCl
(4) NH3
The concentration of which ion is increased when LiOH is dissolved in water?
(1) hydroxide ion
(2) hydrogen ion
(3) hydronium ion
(4) halide ion
Which substance is an Arrhenius acid?
(1) HBr
(2) NaBr
(3) NaOH
(4) NH3
Which pair of compounds represents one Arrhenius acid and one Arrhenius base?
(1) CH3OH and NaOH
(2) CH3OH and HCl
(3) HNO3 and NaOH
(4) HNO3 and HCl
Which type of substance yields hydrogen ions, H+, in an aqueous solution?
(1) an Arrhenius acid
(2) an Arrhenius base
(3) a saturated hydrocarbon
(4) an unsaturated hydrocarbon
Based on the Arrhenius theory, when potassium hydroxide dissolves in water, the only negative ion in the aqueous solution is
(1) O2− (aq)
(2) OH2− (aq)
(3) H− (aq)
(4) OH− (aq)
Which compound is an Arrhenius base?
(1) CO2
(2) CaSO4
(3) Ca(OH)2
(4) C2H5OH
A NaOH(aq) solution with a pH value of 13 is used to determine the molarity of a HCl(aq) solution. A 10.0-mL sample of the HCl(aq) is exactly neutralized by 16.0 mL of 0.100 M NaOH(aq). During this laboratory activity, appropriate safety equipment was used and safety procedures were followed.
Compare the hydronium ion concentration to the hydroxide ion concentration when the HCl(aq) solution is exactly neutralized by the NaOH(aq) solution.
Allow 1 credit. Acceptable responses include, but are not limited to:
• The hydronium ion concentration is equal to the hydroxide ion concentration.
• The concentrations of H3O+ ions and OH− ions are the same.
In a laboratory activity, a student titrates a 20.0-milliliter sample of HCl(aq) using 0.025 M NaOH(aq). In one of the titration trials, 17.6 milliliters of the base solution exactly neutralizes the acid sample.
Identify the positive ion in the sample of HCl(aq).
Allow 1 credit. Acceptable responses include, but are not limited to:
• hydronium ion
• H3O+
• hydronium
• H+
• hydrogen ion
• H3O+(aq)
• hydrogen
• H+(aq)
• proton
A student is to determine the concentration of an NaOH(aq) solution by performing two different titrations. In a first titration, the student titrates 25.0 mL of 0.100 M H2SO4(aq) with NaOH(aq) of unknown concentration.
In a second titration, the student titrates 25.0 mL of 0.100 M HCl(aq) with a sample of the NaOH(aq). During this second titration, the volume of the NaOH(aq) added and the corresponding pH value of the reaction mixture is measured. The graph below represents the relationship between pH and the volume of the NaOH(aq) added for this second titration.
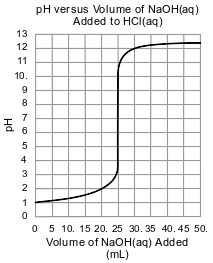
Identify the positive ion present in the H2SO4(aq) solution before the titration.
Allow 1 credit. Acceptable responses include, but are not limited to:
• hydronium ion
• H3O+
• hydronium
• H+
• hydrogen ion
• H3O+(aq)
• hydrogen
• H+ (aq)
• proton
During a titration, 10.00 mL of acetic acid, HC2H3O2(aq), is completely neutralized by adding 12.50 mL of 0.64 M sodium hydroxide, NaOH(aq).
Identify the only positive ion in the HC2H3O2(aq).
Allow 1 credit. Acceptable responses include, but are not limited to:
• H+(aq)
• H3O+
• hydrogen ions
• hydronium
A sample of nitric acid contains both H3O+ ions and NO3− ions. This sample has a pH value of 1. Write a name of the positive ion present in this sample.
Allow 1 credit. Acceptable responses include, but are not limited to: • hydronium ion • hydronium • hydrogen ion • hydrogen
